| Description |
|
Category
|
Registration(Veterinary drug importation)
|
|
Responsible Agency
|
Name: General Authority for Veterinary Services
Address: Mongolia, Ulaanbaatar 13381
Bayanzurkh district,
Peace avenue 16a,
Government building IXa
E-mail: vetsermongolia@magicnet.mn info@dvab.gov.mn
Web: http://vet.gov.mn/
Phone: 51-262271
Fax: 51-263237
|
|
Legal base of the Procedure
|
Law on Business Licensing, 2001
|
|
Time to be spent
|
Resolved within 3 months of receipt of application Issuance of registration certificate within 14 days after the decision to register the drug
|
|
Fee for registration
|
The registration fee (hereinafter referred to as the "registration fee") shall be paid regardless of the form of ownership. The registrant shall be responsible for the costs of work and services related to the registration of drugs, bio-preparations, disinfectants and feed additives. The registration fee shall not be refunded.
|
|
Registration and service fee:
|
- 250,000 tugrugs for the registration of imported drugs, bio-preparations and substances for vaccination, deworming, washing, processing, disinfection and disinfection for animal diseases, and 150,000 tugrugs for renewal;
- Registration of other imported drugs MNT 200,000, extension MNT 100,000;
- 100,000 MNT for registration and 50,000 MNT for renewal of national industrial drugs;
- 100,000 MNT for registration and renewal of drug raw materials;
- The registration fee for the same drug form registered by more than 8 drug supply organizations is twice as high.
|
Required documents:
|
#
|
Type of Information
|
Note |
|
1
|
Trade agreement and power of attorney with the manufacturer
|
|
|
2
|
Pharmacopoeia article
|
|
|
3
|
Documents issued in accordance with the application form for drug registration in the state registration specified in paragraph 8 of this regulation; (must be written and certified in Mongolian)
|
|
|
4
|
Profile of the pharmaceutical company and proof that the plant meets international standards and quality requirements (GMP);
|
|
|
5
|
Test results from an authorized laboratory in the country of origin of the drug or in its own country;
|
|
|
6
|
Published materials on pharmacology and clinical trials, main agents, excipients, stability test results, drug toxicity, activity, composition, safety, interactions, drug label (certified in Mongolian and English).
|
|
|
7
|
Instructions for use written in accordance with the approved model, which includes the composition of the drug, its chemical properties, indications for use, contraindications, side effects, drug interactions, dosage conditions, and warnings;
|
|
|
8
|
Drug samples;
|
|
|
9
|
Drug registration certificate issued by the competent authority of the country;
|
|
|
10
|
Certificate of origin of the drug / Product certified by the competent authority, certificate of industrial registration and free trade, analysis, etc./;
|
|
|
11
|
When registering drug raw materials in the state registry, the manufacturer's GMP certificate, product certificate, test results, methodology, laboratory test results, the conclusion of the environmental assessment of the state central administrative body in charge of nature and environment, and other relevant materials with translation;
|
|
|
12
|
In addition to the above-mentioned documents for registration of bio preparations, the conclusion of the international reference laboratory test;
|
|
|
13
|
A person wishing to register a product in the State Veterinary Drug Registry shall be a legal entity that officially represents the manufacturer in our country and shall submit the original, documents and materials certified by the seal and stamp to the Veterinary Drug Sub-Council.
|
|
|
14
|
Domestic drug labels are written in Mongolian, and imported drug labels are written in English. Instructions for use of the drug are written in Mongolian.
|
|
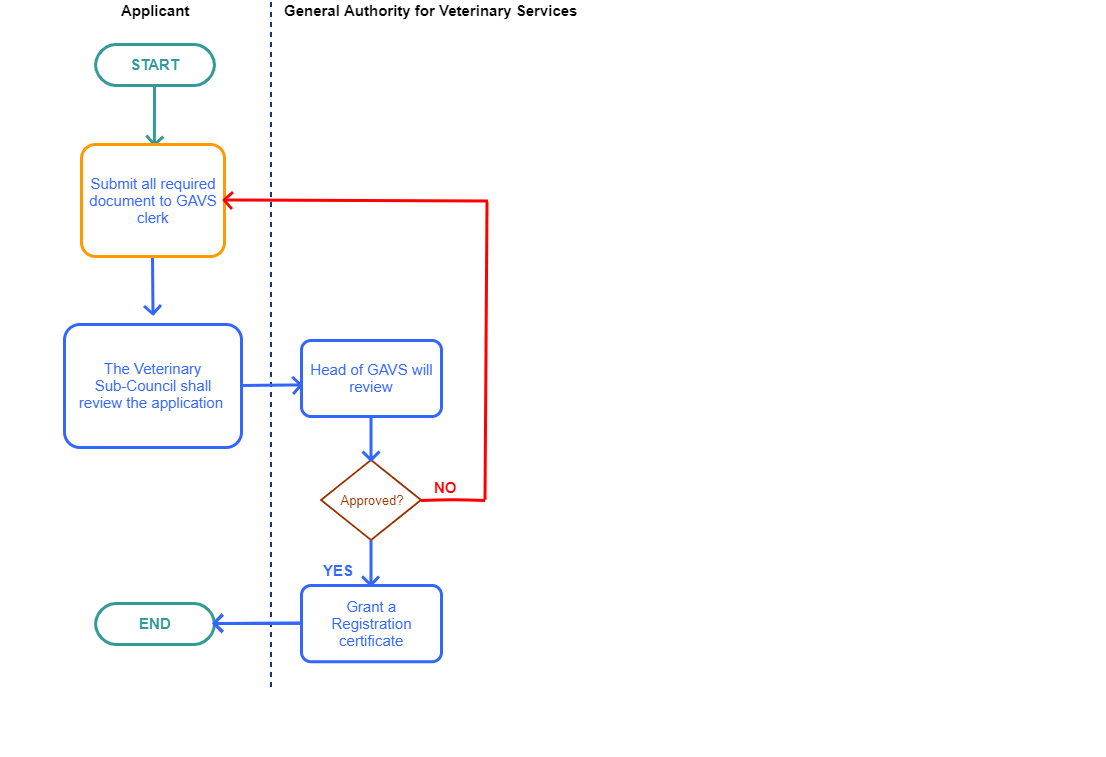
Process step
|
Step 1
|
Submit to the organization's file
|
|
Step 2
|
The Chairman of the Board shall review and approve the application
|
|
Step 3
|
Go to a specialist
The registration certificate will be issued
|
NOTE:
- Drug registration issues shall be resolved within 3 months after receipt of the application materials that meet the requirements specified in 2.3 and 2.4.
- Within 14 days after the decision to register a drug, a registration certificate shall be issued and signed by the chairman and secretary of the drug council.
- The original copy of the registration certificate shall be copied and certified and used for official purposes.
- A copy of the drug certificate of the State Drug Registry for 5 years and a certificate of raw material for 2 years shall be issued to the main registrar and extended for the same period.
- Each registration page for drugs and drug raw materials shall be kept in a certified special confidential book in accordance with the relevant specifications. A file shall be kept for each drug and drug raw material registered in the state registry, and the materials shall be further enriched and used.
- Registered drugs are published on websites and in the press at least twice a year.
- The registration number of a veterinary drug shall include the origin of the drug, the year of registration, the period of registration (by date), the drug classification code (ATCvet), the conditions for issuing the drug, and the personal number.
- Drugs registered in the Veterinary Drug Registry shall be numbered in the format QF-15-0220-A-O-99999
|
|---|


